Engineering Absolute Purity in Viral Clearance
Precision-molded, USP Class VI fluid path components designed for the rigorous demands of nanofiltration and aseptic bioprocessing.
Sterility is not a suggestion—it’s a mandate.
In the high-stakes world of biologics and vaccine manufacturing, the virus filtration stage serves as your final, critical line of defense. Brevet provides the high-performance infrastructure that surrounds your filters, ensuring every transition, split, and connection maintains 100% aseptic integrity. We bridge the gap between high-value drug substances and patient safety with zero-leak, high-purity fluidic engineering that eliminates the risk of bypass or contamination.
The Anatomy of a Filtration Assembly
Our medical-grade polycarbonate components are engineered to withstand the pressures and purity requirements of modern bioprocessing.

Polycarbonate Reducer
Technical Edge: Rigid, dimensionally stable walls.
Primary Application: Facilitates smooth transitions from large process feed lines to sensitive filter inlets without pressure-induced tubing collapse.
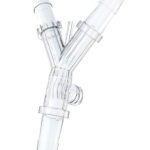
60° Y-Connector
Technical Edge: Optimized internal geometry for laminar flow.
Primary Application: Strategically splits streams into parallel filter banks to increase throughput while minimizing shear stress on delicate proteins.

Perfusion Adapter
Technical Edge: Multi-port, high-density connectivity.
Primary Application: Essential for continuous downstream processing, inline integrity testing, and complex manifold configurations.

Straight Connector
Technical Edge: Micro-tolerance molding for a perfect seal.
Primary Application: Secure, leak-proof inline connections for high-pressure nanofiltration assemblies and final collection paths.
THE BREVET FLUID PATH ADVANTAGE
Optimized Flow from Inlet to Collection
- Inlet: Smooth transition via Brevet Reducers ensures consistent pressure.
- Filtration: High-pressure tolerance in rigid PC housings maintains filter integrity.
- Diversion: 60° Y-Connectors provide low-shear routing for sensitive biologics.
- Collection: Sterile straight connectors ensure zero-leak final product capture.
WHY ENGINEERS CHOOSE BREVET
Precision-Engineered Compliance for the Most Demanding Biological Applications
- Material Excellence: Every component is molded from USP Class VI Medical Grade Polycarbonate for maximum clarity, strength, and biocompatibility.
- Cleanroom Certified: Manufactured in ISO 13485 certified cleanrooms to eliminate particulate contamination at the source.
- Sterilization Ready: Fully compatible with Gamma Irradiation (up to 50kGy) and Autoclave cycles without compromising structural integrity.
- Customization Expertise: Beyond our standard catalog, our engineers partner with OEMs to design bespoke fluid paths for proprietary filtration systems.
